Integrated Therapies for Alcohol use in Alcohol-associated Liver Disease (ITAALD) trial
ITAALD is a multicenter, randomized, double-blinded, placebo-controlled trial focused on the treatment of severe alcohol-associated hepatitis (sAH) and alcohol use disorder (AUD).
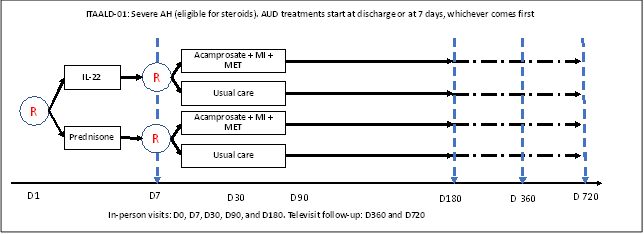
The primary objective of the study is to determine whether subjects receiving F-652 (recombinant IL-22) followed by enhanced AUD treatments will have better alcohol and liver-related outcomes at 6 months compared to those receiving prednisone plus usual care for AUD. Patients assigned to the AUD treatment will receive Acamprosate and counseling, while those assigned to the AUD usual care will receive brief advice and referral to a 12-step program.
The design of the ITAALD trial is shown in the following diagram.
More information about ITAALD